| Name of study |
Confirmation study of the effect of HYA-containing supplement intake on postprandial blood glucose AUC values and postprandial blood glucose levels in subjects with elevated postprandial blood glucose levels (a randomized, placebo-controlled, double-blind, crossover study) |
| Objectives |
To study the effect of HYA-containing supplement on postprandial blood glucose in men and women aged 20-69 years. |
Investigational
product |
Investigational
product |
HYA-containing
supplement |
HYA 50%、LA20%、OA6%、other24% |
Related
components |
10-hydroxy-cis-12-octadecenoic acid (HYA;C18H34O3) |
Form,
packaging |
Capsules, aluminum sachets |
| Intake dose |
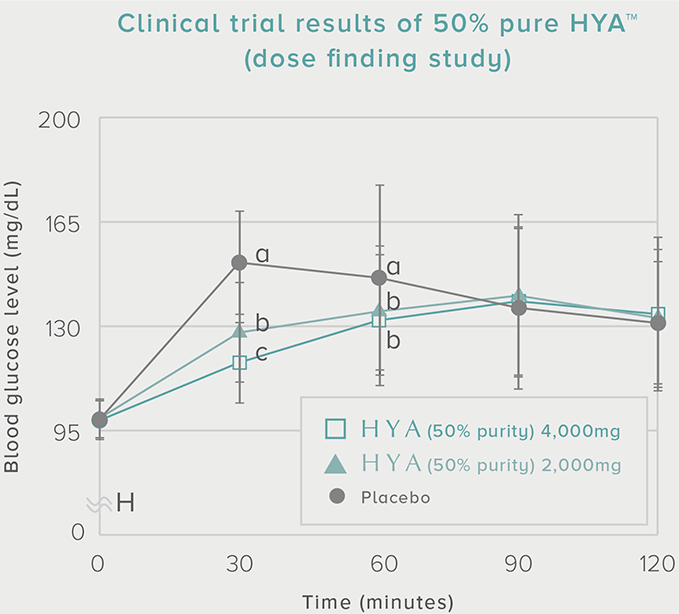
Dose-finding study
1 sachet (200 capsules) once daily
0 mg HYA (placebo)
1,000 mg HYA (2,000 mg of 50% pure HYA)
2,000 mg HYA (4,000 mg of 50% pure HYA) |
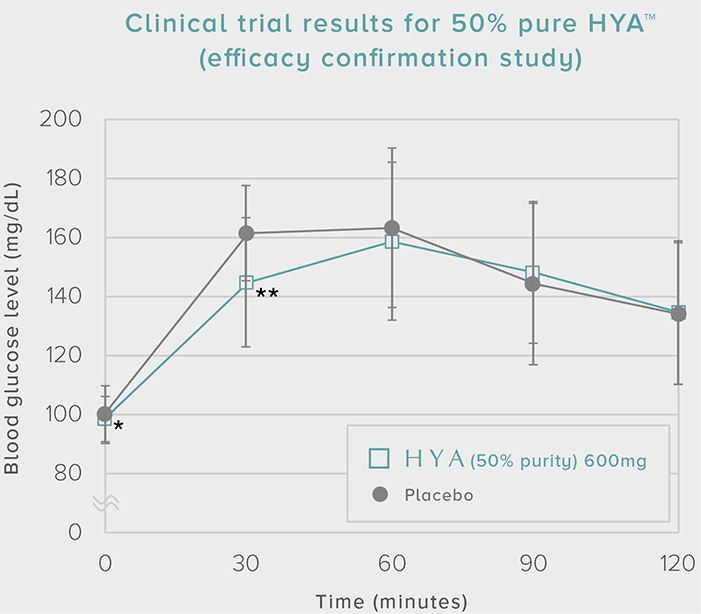
Efficacy confirmation study
One packet (30 capsules) once daily
0 mg HYA (placebo)
300 mg HYA (600 mg of 50% pure HYA) |
| Subjects |
Number of
subjects |
60 subjects (20 x 3 groups) Placebo-controlled crossover |
60 subjects (30 subjects x 2 groups) Placebo-controlled crossover |
Inclusion
criteria |
Subjects with a blood glucose level of (140-199 mg/dL) 30 minutes after a meal at the time of the pre-test. |
| Methods |
Endpoint |
Area under the concentration-time curve for blood glucose level (AUC) and peak postprandial blood glucose level (Cmax) |
Period of
implementation |
July 2016~January 2017 |
April 2017~February 2018 |
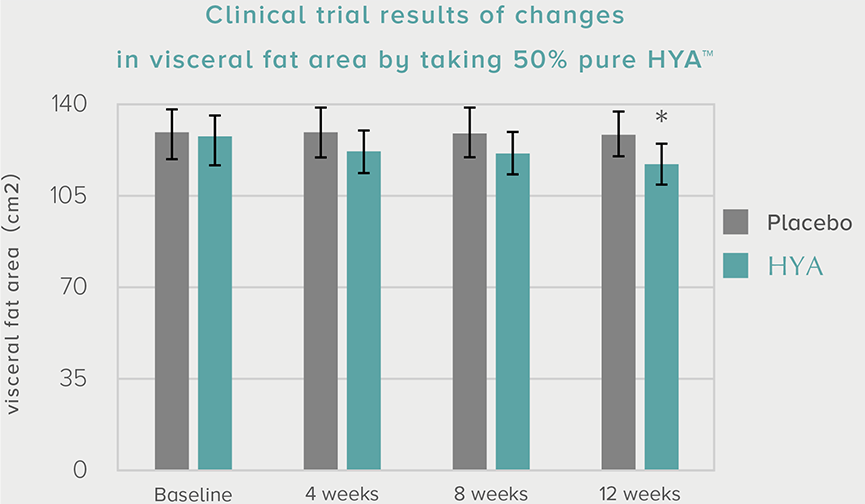
References based on "Progress in Medicine, 37, 1105 -1111 (2017) , Progress in Medicine, 39, 1147-1154 (2019)"
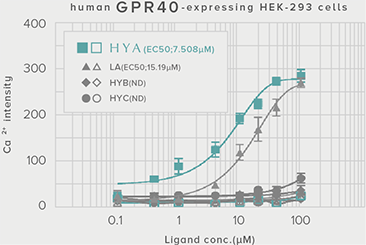
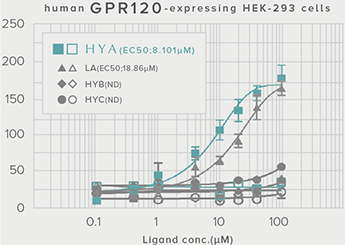
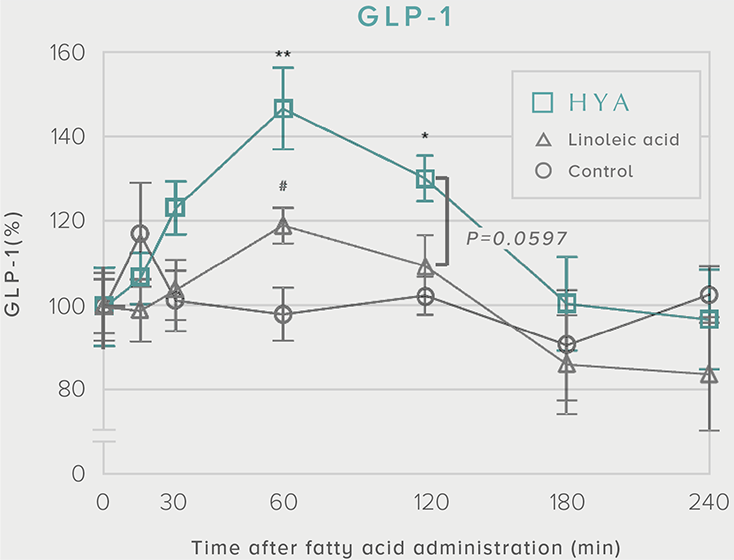
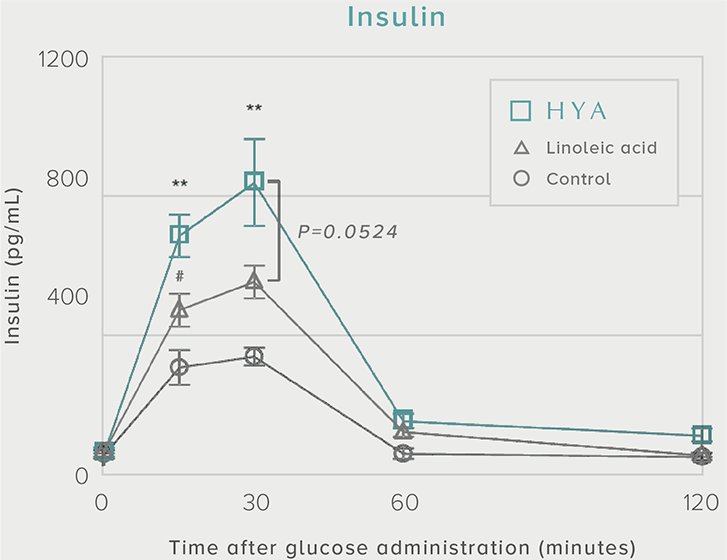
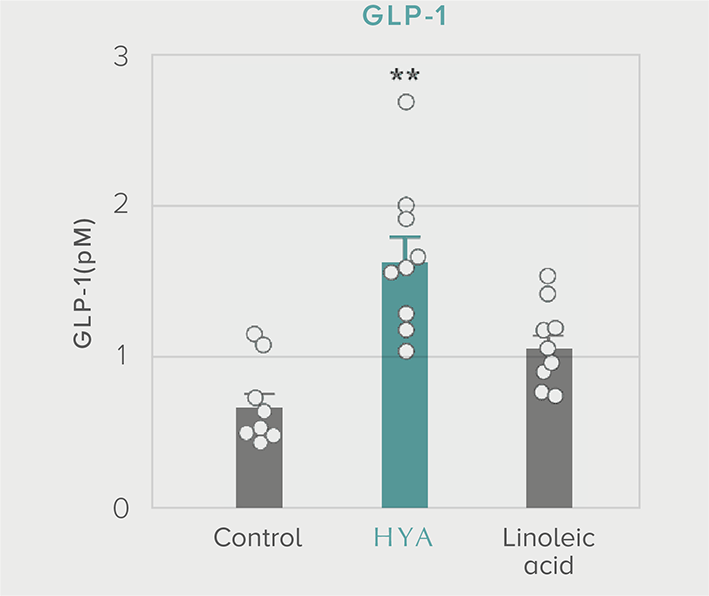
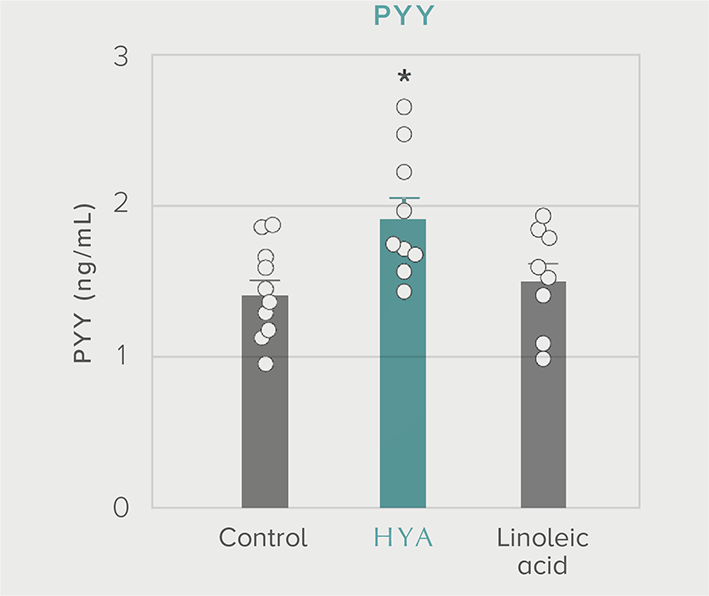
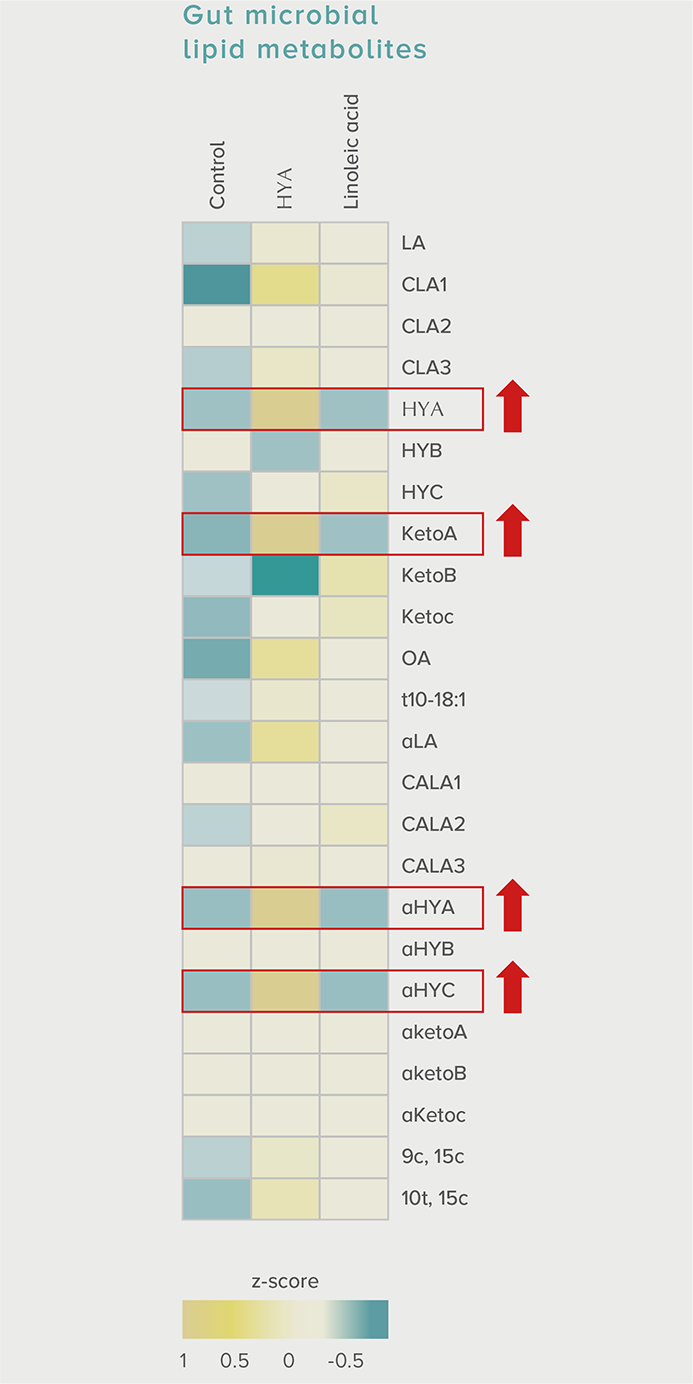
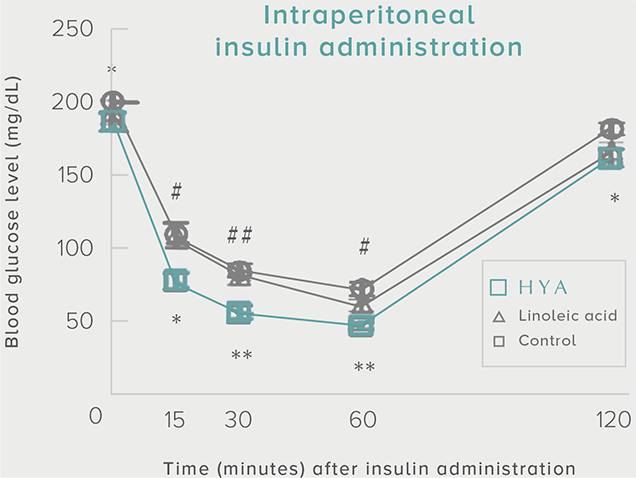
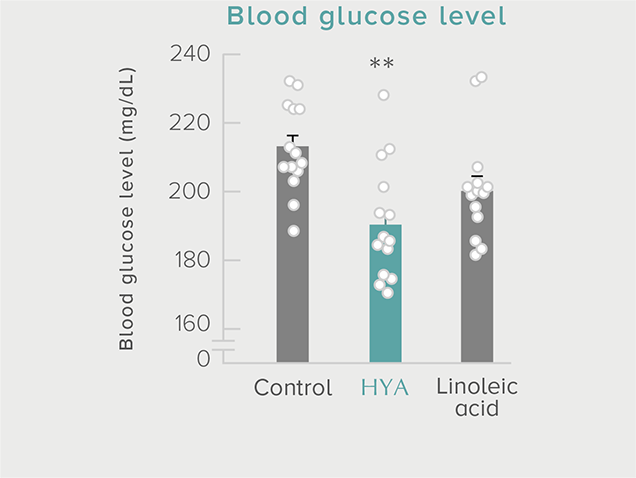
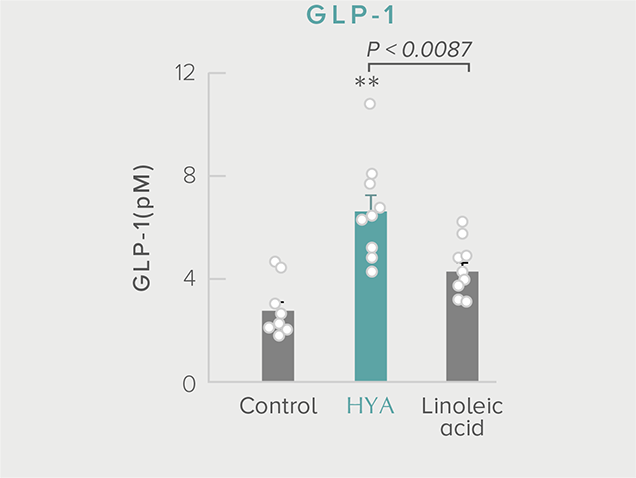
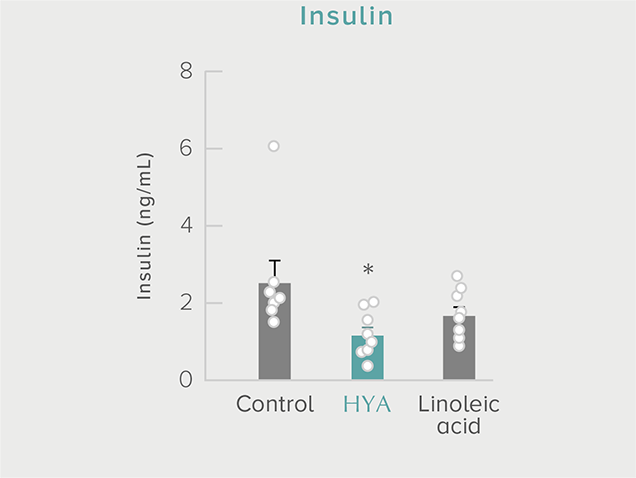
GLP-1 and insulin in reducing blood glucose levels
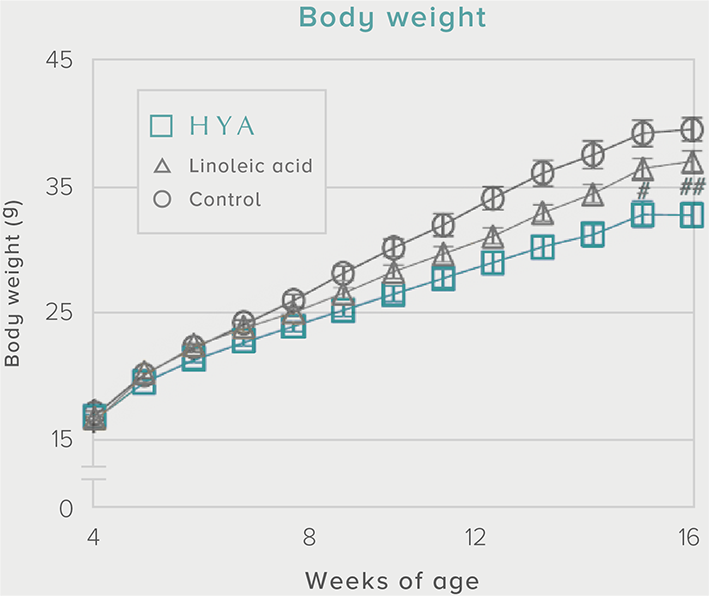
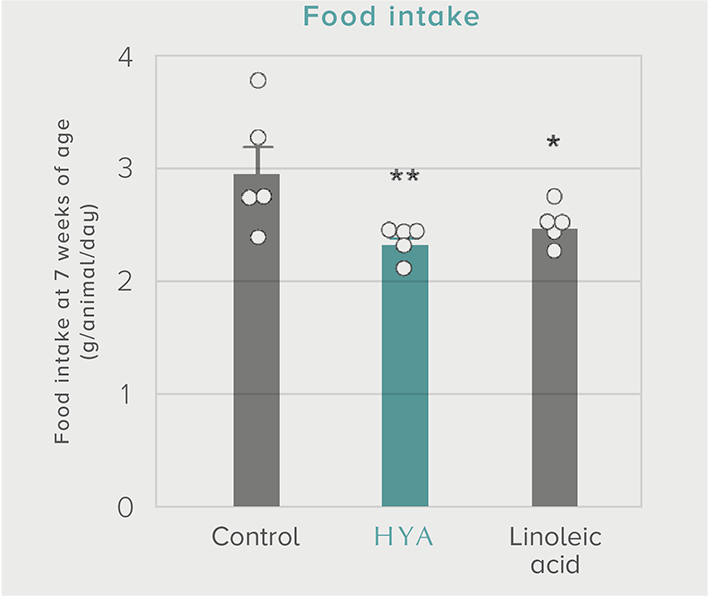
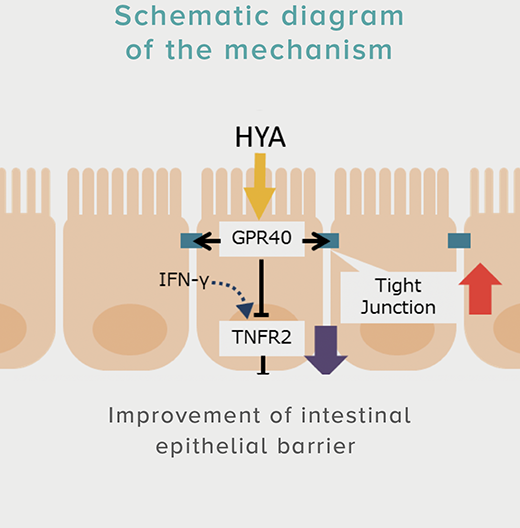
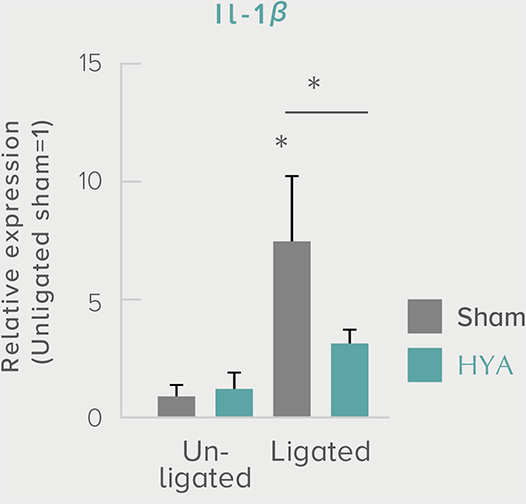
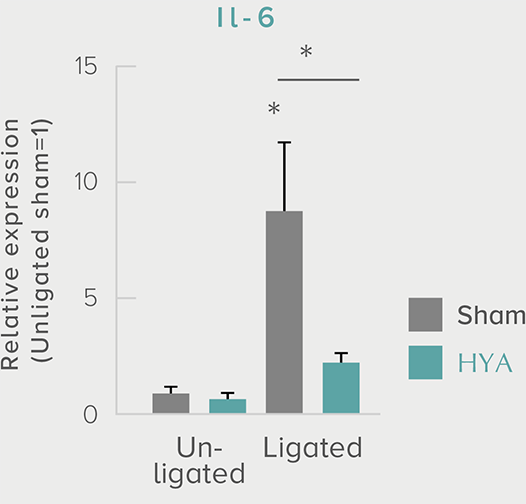
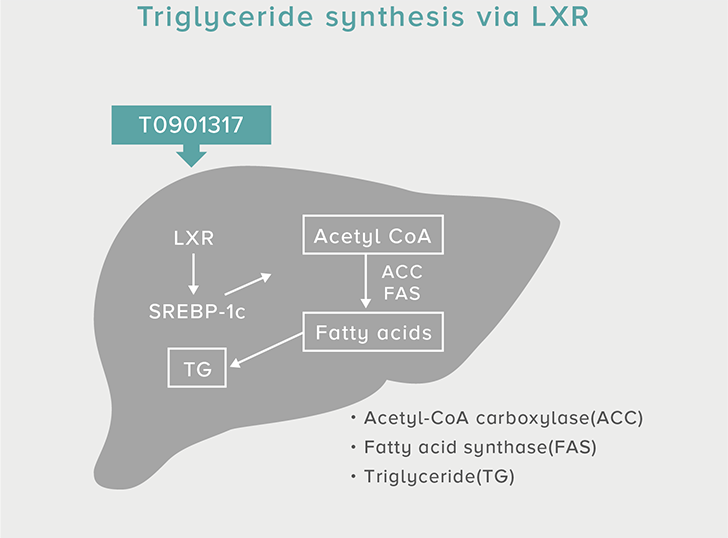
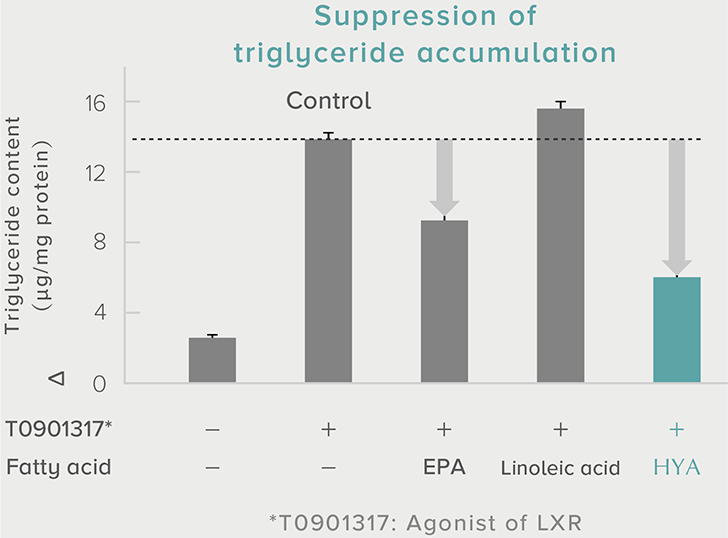
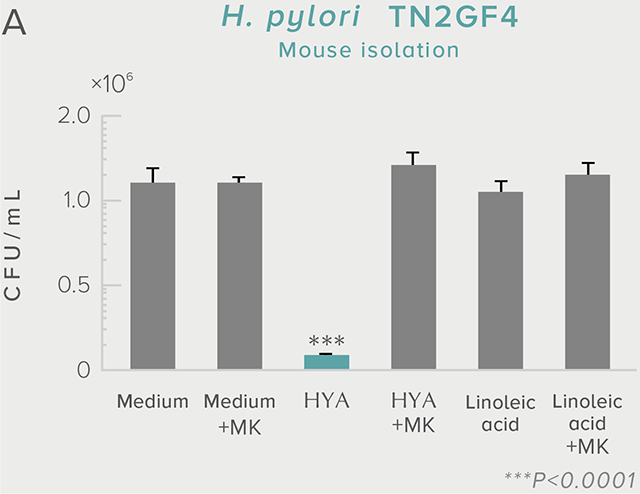
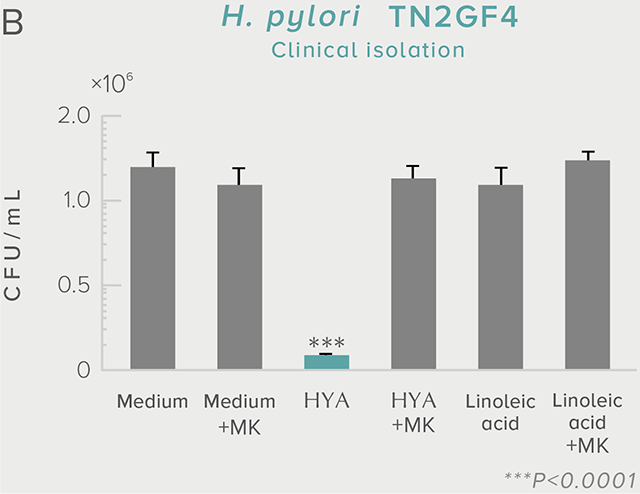
A single dose of HYA promotes GLP-1 secretion and increases insulin levels and it has been confirmed in a study using mice that HYA suppresses the increase in postprandial blood glucose levels.
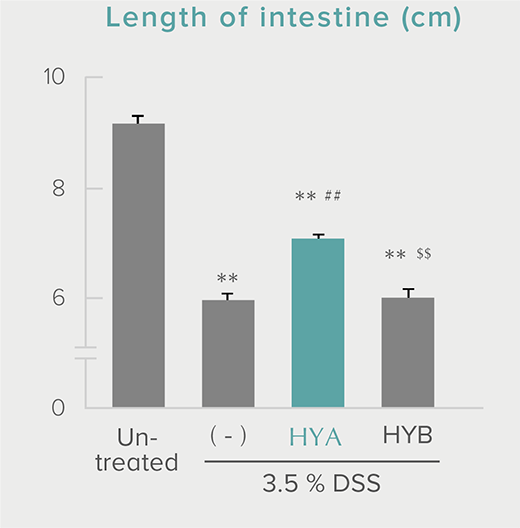
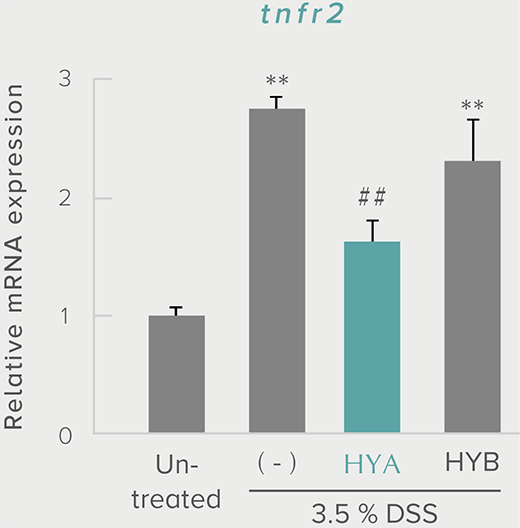
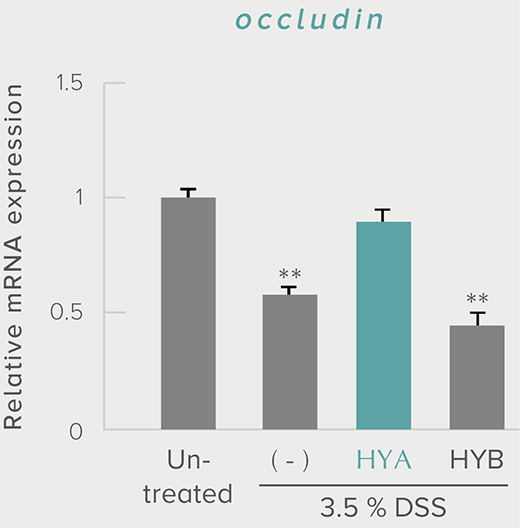
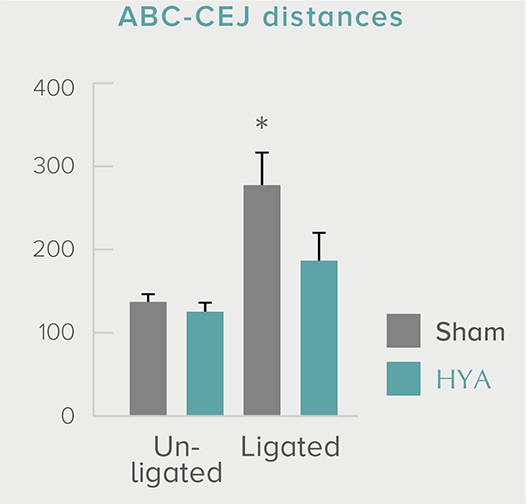
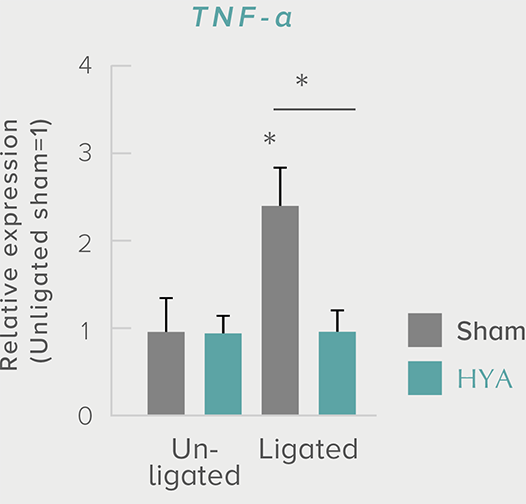
**P < 0.01; *P < 0.05 vs. control (Tukey–Kramer test). ##P <0.01; #P < 0.05 vs. HYA (Tukey–Kramer test).
| Evaluation system |
Mouse (C57BL/6L) |
| Grouping |
HYA group (1,000 mg/kg)
LA group (1,000 mg/kg)
Control group |
| Administration method |
Single oral administration |
References prepared based on "Nature Communications. 10,4007 (2019)".